Scientific Assessment of Ozone Depletion: 2010
Executive Summary

It has been recognized since the 1970s that a number of compounds emitted by human activities deplete stratospheric ozone. The Montreal Protocol on Substances that Deplete the Ozone Layer was adopted in 1987 to protect global ozone and, consequently, protect life from increased ultraviolet (UV) radiation at Earth's surface. Chlorine- and bromine-containing substances that are controlled by the Montreal Protocol are known as ozone-depleting substances (ODSs). ODSs are responsible for the depletion of stratospheric ozone observed in polar regions (for example, the "ozone hole" above Antarctica) and in middle latitudes. The severe depletion of stratospheric ozone observed in the Antarctic has increased UV at the surface and affected climate at southern high latitudes.
The Montreal Protocol and its Amendments and Adjustments have successfully controlled the global production and consumption of ODSs over the last two decades, and the atmospheric abundances of nearly all major ODSs that were initially controlled are declining. Nevertheless, ozone depletion will continue for many more decades because several key ODSs last a long time in the atmosphere after emissions end.
In contrast to the diminishing role of ODSs, changes in climate are expected to have an increasing influence on stratospheric ozone abundances in the coming decades. These changes derive principally from the emissions of long-lived greenhouse gases, mainly carbon dioxide (CO2), associated with human activities. An important remaining scientific challenge is to project future ozone abundances based on an understanding of the complex linkages between ozone and climate change.
Most ODSs are potent greenhouse gases. The buildup of ODS abundances over the last decades contributes to global warming. The actions taken under the Montreal Protocol have reduced the substantial contributions these gases would have made to global warming.
There is now new and stronger evidence of the effect of stratospheric ozone changes on Earth's surface climate, and of the effects of climate change on stratospheric ozone. These results are an important part of the new assessment of the depletion of the ozone layer presented here.
Changes in Gases that Affect Stratospheric Ozone and climate
Changes in the global atmospheric abundance of a substance are determined by the balance between its emissions and removals from the atmosphere. Declines observed for ozone-depleting substances controlled under the Montreal Protocol are due to global emission reductions that have made emissions smaller than removals. Most ODSs are potent greenhouse gases. As the majority of ODSs have been phased out, demand for hydrochlorofluorocarbon (HCFC) and hydrofluorocarbon (HFC) substitutes for the substances controlled under the Montreal Protocol has increased; these are also greenhouse gases. HCFCs deplete much less ozone per kilogram emitted than chlorofluorocarbons (CFCs), while HFCs are essentially non-ozone depleting gases.
Ozone-Depleting Substances and Substitutes: Tropospheric Abundances and Emissions
- The amended and adjusted Montreal Protocol continues to be successful at reducing emissions (Figure ES-1) and thereby abundances of most controlled ozone-depleting substances in the lower atmosphere (troposphere), as well as abundances of total chlorine and total bromine from these ozone-depleting substances. By 2008, the total tropospheric abundance of chlorine from ODSs and methyl chloride had declined to 3.4 parts per billion (ppb) from its peak of 3.7 ppb. However, the rate of decline in total tropospheric chlorine by 2008 was only two-thirds as fast as was expected. This is because HCFC abundances increased more rapidly than expected, while CFCs decreased more slowly than expected. The discrepancy in CFC decreases is most likely because of emissions from "banks" in existing applications such as refrigerators, air conditioners, and foams. The rapid HCFC increases are coincident with increased production in developing countries, particularly in East Asia. The rate of decline of total tropospheric bromine from controlled ODSs was close to that expected and was driven by changes in methyl bromide.
- Declines in CFCs made the largest contribution to the observed decrease in total tropospheric chlorine during the past few years and are expected to continue to do so through the rest of this century. Observations show that CFC-12 tropospheric abundances have decreased for the first time. The decline of methyl chloroform (CH3CCl3) abundances made a smaller contribution to the decrease in total chlorine than described in past Assessments, because this short-lived substance has already been largely removed from the atmosphere.
- Carbon tetrachloride (CCl4) tropospheric abundances have declined less rapidly than expected. Emissions derived from data reported to the United Nations Environment Programme (UNEP) are highly variable and on average appear smaller than those inferred from observed abundance trends. Although the size of this discrepancy is sensitive to uncertainties in our knowledge of how long CCl4 persists in the atmosphere (its "lifetime"), the variability cannot be explained by lifetime uncertainties. Errors in reporting, errors in the analysis of reported data, and/or unknown sources are likely responsible for the year-to-year discrepancies.
- Observations near the tropical tropopause suggest that several very short-lived industrial chlorinated chemicals, not presently controlled under the Montreal Protocol (e.g., methylene chloride, CH2Cl2; chloroform, CHCl3; 1,2 dichloroethane, CH2ClCH2Cl; perchloroethylene, CCl2CCl22), reach the stratosphere. However, their contribution to stratospheric chlorine loading is not well quantified.
- Bromine from halons stopped increasing in the troposphere during 2005-2008. As expected, abundances of halon-1211 decreased for the first time during 2005-2008, while halon-1301 continued to increase but at a slower rate than in the previous Assessment.
- Tropospheric methyl bromide abundances continued to decline during 2005-2008, as expected due to reductions in industrial production, consumption, and emission. About half of the remaining methyl bromide consumption was for uses not controlled by the Montreal Protocol (quarantine and pre-shipment applications).
- Tropospheric abundances and emissions of some HCFCs are increasing faster now than four years ago. Abundances of HCFC-22, the most abundant HCFC, increased more than 50% faster in 2007-2008 than in 2003-2004, while HCFC-142b abundances increased about twice as fast as in 2003-2004. HCFC-141b abundances increased at a similar rate to that observed in 2003-2004. Total emissions of HCFCs are projected to begin to decline during the coming decade due to measures already agreed to under the Montreal Protocol (Figure ES-1).
- Tropospheric abundances and emissions of HFCs, used mainly as substitutes for CFCs and HCFCs, continue to increase. For example, abundances of HFC-134a, the most abundant HFC, have been increasing by about 10% per year in recent years. Abundances of other HFCs, including HFC-125, -143a, -32, and -152a, have also been increasing. Regional studies suggest significant HFC emissions from Europe, Asia, and North America.
Figure ES-1. Emissions of ODSs and their substitutes.
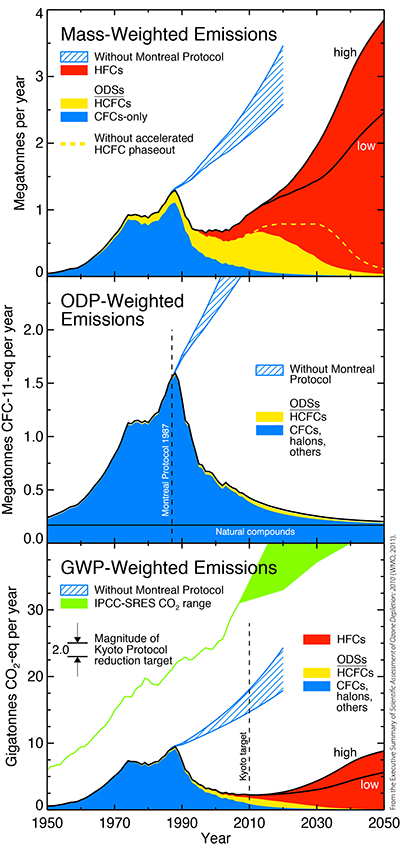
CFCs, HCFCs, HFCs, and Climate Change
- The Montreal Protocol and its Amendments and Adjustments have made large contributions toward reducing global greenhouse gas emissions (Figure ES-1). In 2010, the decrease of annual ODS emissions under the Montreal Protocol is estimated to be about 10 gigatonnes of avoided CO2-equivalent[1] emissions per year, which is about five times larger than the annual emissions reduction target for the first commitment period (2008-2012) of the Kyoto Protocol.
- The sum of the HFCs currently used as ODS replacements contributes about 0.4 gigatonnes of CO2-equivalent per year to total global CO2-equivalent emissions, while the HCFCs contribute about 0.7 gigatonnes. CO2-equivalent emissions of HFCs are increasing by about 8% per year and this rate is expected to continue to grow, while the contribution from HCFCs is expected to start decreasing in the next decade.
- Emissions of HFC-23, a by-product of HCFC-22 production, contributed about 0.2 gigatonnes of CO2-equivalent per year in 2006-2008. HFC-23 is a particularly potent greenhouse gas with a lifetime of about 220 years. Its emissions have increased in the past decade despite global emissions reduction measures, including those covered by the Kyoto Protocol's Clean Development Mechanism projects.
Total Chlorine and Bromine and Implications for Ozone Depletion
- Total chlorine has continued to decline from its 1990s peak values in both the troposphere and the stratosphere. Total tropospheric bromine is decreasing from its peak values, which occurred comparatively recently, while stratospheric bromine is no longer increasing.
- Relative declines in the sum of stratospheric chlorine and bromine from peak values are largest in midlatitudes and smallest in Antarctica (refer to Figure S1-1 in the Scientific Summary of Chapter 1 of this Assessment). These declines are not as pronounced as observed in their tropospheric abundances. Differences between declines in the troposphere and different regions of the stratosphere are primarily associated with the time required for air to move from the troposphere to those regions. The relative declines are smallest in Antarctica primarily because the transport times to polar regions are the largest.
Ozone and Climate: Antarctic
The Antarctic ozone hole is the clearest manifestation of the effect of ODSs on the ozone layer. The depletion far exceeds natural variability and has occurred without exception since 1980. The ozone hole also provides the most visible example of how ozone depletion affects surface climate.
Figure ES-2. Schematic of the influence of ozone-depleting substances (ODSs) and climate change on the stratospheric ozone layer, and the influence of ozone changes on surface ultraviolet radiation.
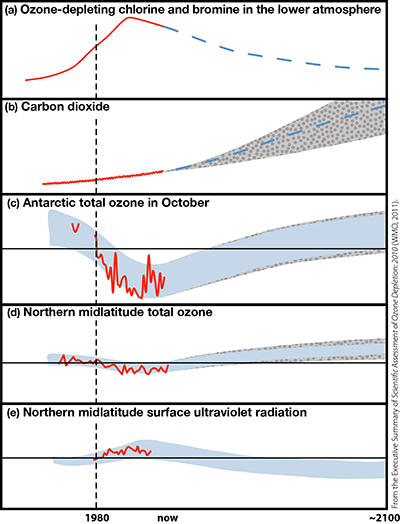
- Springtime Antarctic total column ozone losses (the ozone hole), first recognizable around 1980, continue to occur every year (Figure ES-2c). Although the ozone losses exhibit year-to-year variations that are primarily driven by year-to-year changes in meteorology, October mean column ozone within the vortex has been about 40% below 1980 values for the past fifteen years. The average erythemal ("sunburning") UV measured at the South Pole between 1991 and 2006 was 55-85% larger than the estimated values for the years 1963-1980.
- Doubts raised since the previous Assessment regarding our understanding of the cause of the Antarctic ozone hole have been dispelled. New laboratory measurements on the key chemistry involved in polar ozone depletion have reaffirmed that past changes in ODSs are indeed the cause of the ozone hole. This is also supported by quantification of the chemicals responsible for the ozone hole via field observations.
- There is increased evidence that the Antarctic ozone hole has affected the surface climate in the Southern Hemisphere. Climate models demonstrate that the ozone hole is the dominant driver of the observed changes in surface winds over the Southern Hemisphere mid and high latitudes during austral summer. These changes have contributed to the observed warming over the Antarctic Peninsula and cooling over the high plateau. The changes in the winds have also been linked to regional changes in precipitation, increases in sea ice around Antarctica, warming of the Southern Ocean, and a local decrease in the ocean sink of CO2.
- The trends in the summertime winds in the Southern Hemisphere are not expected to persist over the next few decades. This is because of the expected offsetting influences on the surface winds of increasing greenhouse gases and the recovering ozone hole.
- Observed Antarctic springtime column ozone does not yet show a statistically significant increasing trend (Figure ES-2c). Year-to-year variability, due to meteorology, is much larger than the expected response to the small ODS decreases in the Antarctic vortex to date. This is consistent with simulations using chemistry-climate models (CCMs).
- The evolution of Antarctic springtime column ozone over the rest of the century is expected to be dominated by the decline in ODS abundance (Figure ES-2c). CCM simulations show that greenhouse gas changes have had, and will continue to have, a small impact on the ozone hole compared to the effects of the ODS changes. There are some indications that small episodic Antarctic ozone holes may occur even through the end of the century. In spring and early summer, Antarctica will continue to experience excess surface UV.
Ozone and Climate: Global and Arctic
As a result of the controls introduced by the Montreal Protocol and its Amendments and Adjustments, it is expected that the decline in ODSs will lead to an increase in stratospheric ozone abundances. However, it will be challenging to attribute ozone increases to the decreases in ODSs during the next few years because of natural variability, observational uncertainty, and confounding factors, such as changes in stratospheric temperature or water vapor. A feature of this Assessment is the coordinated use by the community of chemistry-climate models (CCMs) with integrations covering the period from 1960-2100, which has allowed more detailed study of the long-term changes in the stratosphere and of the relative contributions of ODSs and greenhouse gases (GHGs).
- Average total ozone values in 2006-2009 remain at the same level as the previous Assessment, at roughly 3.5% and 2.5% below the 1964-1980 averages respectively for 90°S-90°N and 60°S-60°N. Midlatitude (35°-60°) annual mean total column ozone amounts in the Southern Hemisphere [Northern Hemisphere] over the period 2006-2009 have remained at the same level as observed during 1996-2005, at ~6% [~3.5%] below the 1964-1980 average.
- The ozone loss in Arctic winter and spring between 2007 and 2010 has been variable, but has remained in a range comparable to the values prevailing since the early 1990s. Substantial chemical loss continues to occur during cold Arctic winters.
- Robust linkages between Arctic stratospheric ozone depletion and tropospheric and surface climate trends have not been established, as expected from the smaller ozone depletion compared with the Antarctic.
- Chemistry-climate models reproduce both the latitudinal and vertical structure of the observed ozone trends in both northern and southern midlatitudes during the past periods of increase of the ODSs, confirming our basic understanding of ozone change. Simulations agree with observations that the last decade has shown flattening of the time series of global total ozone.
- Analyses based on surface and satellite measurements show that erythemal UV irradiance over midlatitudes has increased since the late 1970s (Figure ES-2e). This is in qualitative agreement with the observed decrease in column ozone, although other factors (mainly clouds and aerosols) have influenced long-term changes in erythemal irradiance. Clear-sky UV observations from unpolluted sites in midlatitudes show that since the late 1990s, UV irradiance levels have been approximately constant, consistent with ozone column observations over this period.
- New analyses of both satellite and radiosonde data give increased confidence in changes in stratospheric temperatures between 1980 and 2009. The global-mean lower stratosphere cooled by 1-2 K and the upper stratosphere cooled by 4-6 K between 1980 and 1995. There have been no significant long-term trends in global-mean lower stratospheric temperatures since about 1995. The global-mean lower-stratospheric cooling did not occur linearly but was manifested as downward steps in temperature in the early 1980s and the early 1990s. The cooling of the lower stratosphere includes the tropics and is not limited to extratropical regions as previously thought.
- The evolution of lower stratospheric temperature is influenced by a combination of natural and human factors that has varied over time. Ozone decreases dominate the lower stratospheric cooling since 1980. Major volcanic eruptions and solar activity have clear shorter-term effects. Models that consider all of these factors are able to reproduce this temperature time history.
- Changes in stratospheric ozone, water vapor, and aerosols all radiatively affect surface temperature. The radiative forcing[2] of climate in 2008 due to stratospheric ozone depletion (-0.05 ± 0.1 Watts per square meter, W/m2) is much smaller than the positive radiative forcing due to the CFCs and HCFCs largely responsible for that depletion (about +0.3 W/m2). For context, the current forcing by CO2 is approximately +1.7 W/m2. Radiative calculations and climate modeling studies suggest that the radiative effects of variability in stratospheric water vapor (± ~0.1 W/m2 per decade) can contribute to decadal variability in globally averaged surface temperature. Climate models and observations show that major volcanic eruptions (e.g., Mt. Pinatubo in 1991, roughly -3 W/m2) can cool the surface for several years.
- The global middle and upper stratosphere are expected to cool in the coming century, mainly due to CO2 increases. Stratospheric ozone recovery will slightly offset the cooling. HFCs could warm the tropical lower stratosphere and tropopause region by about 0.3°C if stratospheric abundances reach the 1 ppb level.
- Emerging evidence from model simulations suggests that increasing greenhouse gases lead to an acceleration of the stratospheric circulation usually referred to as the Brewer-Dobson circulation. Such an acceleration could have important consequences, particularly decreases in column ozone in the tropics and increases in column ozone elsewhere. However, responsible mechanisms remain unclear and observational evidence for the circulation increase is lacking.
- Global ozone is projected to increase approximately in line with the ODS decline, and the increase is accelerated by cooling of the upper stratosphere. Global ozone is not very sensitive to circulation changes, so high confidence can be placed in this projection.
- The evolution of ozone in the Arctic is projected to be more sensitive to climate change than in the Antarctic. The projected strengthening of the stratospheric Brewer-Dobson circulation is expected to significantly increase lower stratospheric ozone in the Arctic, augmenting the GHG-induced ozone increase from upper stratospheric cooling and hastening the return to 1980 levels.
- GHG-induced temperature and circulation changes are projected to hasten the return of midlatitude total column ozone to 1980 levels by several decades, rising well above 1980 levels by the end of the century. The effect is most pronounced in northern midlatitudes (Figure ES-2d), where it would result in clear-sky surface UV radiation levels well below 1980 values by the second half of the century (Figure ES-2e). In southern midlatitudes, the effect of circulation changes is projected to be weaker and ozone is also influenced by depletion in the Antarctic, where the return to 1980 levels occurs much later.
Information for Policymakers and Options for Policy Formulation
Cases related to the elimination of future emissions, production, and banks for various ozone-depleting substances (ODSs) can be formulated starting from a baseline future emission scenario. The baseline scenario here has been developed to account for past and present levels of ODSs along with emission projections. This scenario projects that stratospheric chlorine and bromine levels are likely to return to 1980 levels in midcentury for the midlatitudes and about 25 years later in the Antarctic vortex. These additional cases are used to evaluate the impact of various hypothetical policy options.
Information for Policymakers
- The Montreal Protocol has both protected the ozone layer and provided substantial co-benefits by reducing climate change (see Figure ES-1, bottom two panels). It has protected the stratospheric ozone layer by phasing out production and consumption of ozone-depleting substances. Simulations show that unchecked growth in the emissions of ODSs would have led to global ozone depletion in the coming decades very much larger than current levels. Solar UV radiation at the surface would also have increased substantially.
- Projections of hydrofluorocarbon (HFC) growth in scenarios that assume no controls suggest that by 2050, Global Warming Potential-weighted emissions from these substances could be comparable to the GWP-weighted emissions of chlorofluorocarbons (CFCs) at their peak in 1988 (see Figure ES-1, bottom panel). The highest projection assumes that developing countries use HFCs with GWPs comparable to those currently in use.
- The accelerated hydrochlorofluorocarbon (HCFC) phase-out agreed to by the Parties to the Montreal Protocol in 2007 is projected to reduce ozone depletion and to help reduce climate forcing (see Figure ES-1). This acceleration is expected to reduce cumulative HCFC emissions by about 0.7 million Ozone Depletion Potential-tonnes between 2011 and 2050 and would bring forward the year equivalent effective stratospheric chlorine (EESC) returns to 1980 levels by 4-5 years. The accelerated HCFC phasedown is projected to reduce greenhouse gas emissions by about 0.5 gigatonnes of carbon dioxide (CO2)-equivalent per year averaged over 2011 through 2050. The projected benefit would be determined by the climate impact of the replacements. In comparison, global anthropogenic emissions of CO2 were greater than 30 gigatonnes per year in 2008.
- Since the previous Assessment, new fluorocarbons have been suggested as possible replacements for potent HCFC and HFC greenhouse gases. For example, HFC-1234yf (Ozone Depletion Potential (ODP) = 0; 100-year GWP = 4) is proposed to replace HFC-134a (ODP = 0; 100-year GWP = 1370) in mobile air conditioning. To fully assess the environmental impacts, each proposed substance would need to be evaluated for its ODP, GWP, atmospheric fate, safety, and toxicity. Preliminary analyses indicate that global replacement of HFC-134a with HFC-1234yf at today's level of use is not expected to contribute significantly to tropospheric ozone formation or produce harmful levels of the degradation product TFA (trifluoroacetic acid). It is well established that TFA is a ubiquitous component of the environment, but uncertainties remain regarding its natural and anthropogenic sources, long-term fate, and abundances.
Due to the success of the Montreal Protocol and its Amendments and Adjustments in reducing the production, emissions, and abundances of controlled ODSs, emissions from other compounds and activities not controlled by the Montreal Protocol have become relatively more important to stratospheric ozone.
- Increasing abundances of radiatively important gases, especially carbon dioxide (CO2) and methane (CH4), are expected to significantly affect future stratospheric ozone through effects on temperature, winds, and chemistry. CO2 increased in the atmosphere at 2.1 parts per million per year from 2005-2008, while CH4 increased by about 6.7 parts per billion per year from 2006-2008.
- Nitrous oxide (N2O) is known to both deplete global ozone and warm the climate. The current ODP-weighted anthropogenic emission is larger than that of any ODS.
- Deliberate large injections of sulfur-containing compounds into the stratosphere, which have been suggested as a climate intervention approach (geoengineering), would alter the radiative, dynamical, and chemical state of the stratosphere and could be expected to have substantial unintended effects on stratospheric ozone levels.
Options for Policy Formulation
Additional cases have been developed to show the impact of further control measures on various substances. Table ES-1 shows the percentage reductions in integrated chlorine and bromine levels and integrated GWP-weighted emissions, relative to the baseline scenario, that can be achieved in these hypothetical cases.
- Halons and CFCs: Leakage from banks is the largest source of current ODP-weighted emissions of ODSs. A delay in the capture and destruction of estimated CFC banks from 2011 to 2015 is currently thought to reduce the possible ozone and climate benefits that could be achieved by about 30%.
- Carbon tetrachloride (CCl4): Elimination of future CCl4 emissions after 2010 would have an EESC impact comparable to the capture and destruction of CFC and halon banks. This is a much larger effect than was estimated in the previous Assessment because of a revision in the estimated emissions.
- HCFCs: The recent growth in reported HCFC production in developing countries was larger than projected in the previous Assessment. This alone would have resulted in a larger projected HCFC production in the new baseline scenario compared to the previous Assessment, but is expected to be more than compensated for by the accelerated HCFC phasedown agreed to by the Parties to the Montreal Protocol in 2007.
- Elimination of all emissions of chlorine- and bromine-containing ODSs after 2010: This would bring forward the return of EESC to 1980 levels by about 13 years. The elimination of these ODS emissions would have a climate impact equivalent to about a 0.7 gigatonnes of CO2-equivalent per year reduction from 2011 through 2050, on average. The sum of current banks of CFCs plus HCFCs contributes about the same amount to these CO2-equivalent emissions as future HCFC production.
- Methyl bromide: Two methyl bromide cases were examined. Case 1: A phase-out of quarantine and pre-shipment emissions beginning in 2011 would accelerate the return of EESC to 1980 levels by 1.5 years, relative to a case of maintaining emissions at 2004-2008 average levels. Case 2: Continuing critical-use exemptions at the approved 2011 level indefinitely would delay the return of EESC to 1980 levels by 0.2 year.
Table ES-1. Hypothetical cases.
Reductions in integrated chlorine and bromine levels (as measured by equivalent effective stratospheric chlorine, EESC) and integrated GWP-weighted emissions, relative to the baseline scenario, that can be achieved in hypothetical cases developed to show the impact of further control measures on various substances.
| Substance or Group of Substances | Reductions (%) in Integrated EESC (equivalent effective stratospheric chlorine) | Reduction in Cumulative GWP-Weighted Emissions from 2011 to 2050 (gigatonnes of CO2-equivalent) | ||
|---|---|---|---|---|
| Bank capture and destruction in 2011 and 2015: | 2011 | 2015 | 2011 | 2015 |
| CFCs | 11 | 7.0 | 7.9 | 5.5 |
| Halons | 14 | 9.1 | 0.4 | 0.3 |
| HCFCs | 4.8 | 5.3 1 | 4.9 | 5.5 1 |
| Production elimination after 2010: | ||||
| HCFCs | 8.8 | 13.2 | ||
| CH3Br for quarantine and pre-shipment | 6.7 | 0.002 | ||
| Total emissions elimination after 2010: | ||||
| CCl4 2 | 7.6 | 0.9 | ||
| CH3CCl3 | 0.1 | 0.004 | ||
| HFCs | 0.0 | Up to 170 3 | ||
1 The impact of a 2015 HCFC bank recovery is larger than a 2011 bank recovery because this calculation assumes destruction of the bank in only a single year, and because the bank in 2015 is larger than the bank in 2011 owing to continued annual production that is larger than the annual bank release.
2 Banks are assumed to be zero. Emissions include uncertain sources such as possible fugitive emissions and unintended by-product emissions.
3 Strongly dependent on future projections and does not consider HFC-23 emissions. Currently HFCs are not controlled by the Montreal Protocol, but are included in the basket of gases of the Kyoto Protocol.
[1] GWP-weighted emissions, also known as CO2-equivalent emissions, are defined as the amount of gas emitted multiplied by its 100-year Global Warming Potential (GWP).
[2] Positive radiative forcings generally warm the surface; negative radiative forcings generally cool the surface.